Collaborators:
David Delgado, Hailey Hendriks, Hannah Midles and Rachel Werner made up one group. In addition, the entire class collaborated together to complete the data table for this experiment.
David Delgado, Hailey Hendriks, Hannah Midles and Rachel Werner made up one group. In addition, the entire class collaborated together to complete the data table for this experiment.
Introduction & Problem:
The pH level of a substance refers to the amount of hydrogen (H) ions in that substance, or the acidity; the lower the pH is, the more hydrogen ions there are. For those who possess some erudition about soil health, this would indicate that slightly acidic water is actually beneficial since it promotes plant growth. For this reason, rainwater, which has a pH of approximately 5.6, is auspicious for normal plant growth. Rainwater is slightly acidic at typical atmospheric concentrations of CO2 because of the fact that "CO2 dissolves into rainwater forming weak carbonic acid"(Acid Rain, EPA). On the other hand, acid rain is an anthropogenic phenomenon that is best known for its pernicious effects on fish populations, vegetative growth, soil health and water quality. According to the Environmental Protection Agency (EPA), acid rain consists of precipitation with a pH value of less than about 5.3 due to excess sulfur dioxide (SO2) and nitrogen oxides (NOx) released by both natural sources -- volcanoes and decaying vegetation -- and man-made sources -- industries (i.e. electrical power generation) and transportation that require the combustion of fossil fuels.
Some effects of this abnormally acidic precipitation have already been published by reliable scientists to further augment this research study. It is known that if precipitation becomes too acidic, then alkaline chemicals in the soil may not be able to neutralize all of the acids found in rainwater. "Over time, these neutralizing materials can be washed away by acid rain. Damage to crops, trees, lakes, rivers, and animals can result"(Acid Rain, EPA). Despite having this prior knowledge, testing the effects of pH on plant growth becomes edifying in terms of determining how harmful certain human activities can be to the ambient environment. Furthermore, the development of remediation methods is necessary to replace current methods of mitigating the effects of acid rain on plants: some farmers compensate for the loss of nutrients due to acid rain by adding fertilizers to the soil. They also add crushed limestone -- an alkaline rock -- to the soil to serve as a pH buffer. Thus, the problem that will be addressed throughout this experiment is, "How does acid rain affect the growth of fescue plants?"
Hypothesis:
If fescue plants are exposed to substances with different levels of acidity -- one with a pH level of 5, the other with a pH level of 2.4, and the last with a pH level of 4 --, then the grass exposed to the sample with a pH level of 5 (water only) will experience the most growth and be the healthiest, the grass exposed to the vinegar-water mixture with a pH level of 4 will experience less growth and be less healthy, and the grass exposed to the sample of water with a pH level of 2.4 (vinegar only) will undergo the least growth and be the least healthy of the three fescue plants.
Parts of the Experiment:
Materials & Methods:
a) Materials:
b) Method:
Data & Data Analysis:
a) Data:
The pH level of a substance refers to the amount of hydrogen (H) ions in that substance, or the acidity; the lower the pH is, the more hydrogen ions there are. For those who possess some erudition about soil health, this would indicate that slightly acidic water is actually beneficial since it promotes plant growth. For this reason, rainwater, which has a pH of approximately 5.6, is auspicious for normal plant growth. Rainwater is slightly acidic at typical atmospheric concentrations of CO2 because of the fact that "CO2 dissolves into rainwater forming weak carbonic acid"(Acid Rain, EPA). On the other hand, acid rain is an anthropogenic phenomenon that is best known for its pernicious effects on fish populations, vegetative growth, soil health and water quality. According to the Environmental Protection Agency (EPA), acid rain consists of precipitation with a pH value of less than about 5.3 due to excess sulfur dioxide (SO2) and nitrogen oxides (NOx) released by both natural sources -- volcanoes and decaying vegetation -- and man-made sources -- industries (i.e. electrical power generation) and transportation that require the combustion of fossil fuels.
Some effects of this abnormally acidic precipitation have already been published by reliable scientists to further augment this research study. It is known that if precipitation becomes too acidic, then alkaline chemicals in the soil may not be able to neutralize all of the acids found in rainwater. "Over time, these neutralizing materials can be washed away by acid rain. Damage to crops, trees, lakes, rivers, and animals can result"(Acid Rain, EPA). Despite having this prior knowledge, testing the effects of pH on plant growth becomes edifying in terms of determining how harmful certain human activities can be to the ambient environment. Furthermore, the development of remediation methods is necessary to replace current methods of mitigating the effects of acid rain on plants: some farmers compensate for the loss of nutrients due to acid rain by adding fertilizers to the soil. They also add crushed limestone -- an alkaline rock -- to the soil to serve as a pH buffer. Thus, the problem that will be addressed throughout this experiment is, "How does acid rain affect the growth of fescue plants?"
Hypothesis:
If fescue plants are exposed to substances with different levels of acidity -- one with a pH level of 5, the other with a pH level of 2.4, and the last with a pH level of 4 --, then the grass exposed to the sample with a pH level of 5 (water only) will experience the most growth and be the healthiest, the grass exposed to the vinegar-water mixture with a pH level of 4 will experience less growth and be less healthy, and the grass exposed to the sample of water with a pH level of 2.4 (vinegar only) will undergo the least growth and be the least healthy of the three fescue plants.
Parts of the Experiment:
- The control group consists of the sample of fescue grass that was exposed to the sample A -- the water with a pH level of 5 that consisted of only water.
- The experimental group is constituted by the sample of fescue grass that was exposed to the sample B -- the water with a pH level of 2.4 -- and the sample of fescue grass exposed to sample C -- the vinegar-water mixture with a pH level of 4.
- The independent variable is the varying pH levels of the water samples to which the samples of fescue plants were exposed.
- The dependent variable is the growth and health of the samples of fescue grass when they were exposed to the water with varying pH levels -- sample B and C.
- The controlled variables include the materials, the height at which each sample of fescue grass was cut, the amount of water to which each sample of fescue grass was exposed, the frequency at which each sample of fescue grass was watered, and the humidity of the classroom.
Materials & Methods:
a) Materials:
- 9 2-L soda bottles containing fescue plants
- Potting soil
- Water
- White Vinegar
- Sunlight
- Scissors
- Ruler
- Tape
- Beaker of water with a pH level of 5 (control - water only)
- Beaker of water with a pH level of 2.4 (vinegar only)
- Beaker of water with a pH level of 4 (vinegar/water mixture)
- pH paper
- Paper towels
b) Method:
- Pour 1 teaspoon of vinegar into 2 cups of distilled water, stir well, and check the pH with either pH paper or a garden soil pH testing kit. The pH of the vinegar/water mixture should be about 4.
- If it is below pH 4, add a sprinkle of baking soda, or a drop of ammonia, stir well, and recheck the pH.
- If it is above pH 4, add a drop or two of vinegar and again recheck the pH.
- Measure the pH of the distilled water using either pH paper.
- Create a second pH solution of your choice. Record the pH of this solution.
- If the pH is below 7, add about 1/8 teaspoon baking soda, or a drop of ammonia, stir well, and check the pH of the water with the pH indicator.
- If the water is still acidic, repeat the process until pH 7 is reached. Should you accidentally add too much baking soda or ammonia, either start over again or add a drop or two of vinegar, stir, and recheck the pH.
- Each group will be responsible for watering 1 plant one day a week for a total of three weeks according to their sample of water.
- Record qualitative and quantitative data throughout the duration of the experiment in Table 1.
Data & Data Analysis:
a) Data:
b) Data Analysis:
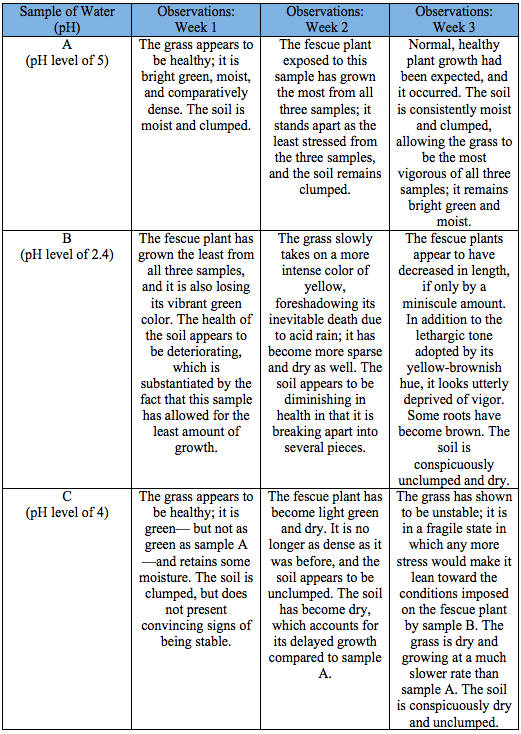
The experiment began with an attempt to make all three of the fescue plants indistinguishable. They were cut down to a length of 6 cm, and had been watered the same amount prior to this experiment. Picture 1 shows the condition of the three samples of grass (samples A, B, and C) before they were exposed to their corresponding samples of water once a week for a total of three weeks. Then, the experiment was conducted, and the data collected. After collecting the aforementioned data, it became apparent that acid rain has pernicious effects on plant growth and soil health; this was further substantiated by the fact that soil exposed to the water with a pH level of 5 (sample A) sustained an aesthetic appearance -- moist and clumped --, while the soil exposed to the water with a pH level of 2.4 (sample B) became dry and unclumped. Sample B resulted in decreased plant growth, and it expedited the falling apart of the soil particles. The fescue plant exposed to the water with a pH level of 4 (sample C) had characteristics similar to sample B in terms of the conditions of the soil and plant growth, which would indicate that an inverse correlation exists between the acidity of water to which the grass is exposed and vegetative growth.
A cursory glance at the three fescue plants would testify to the fact that the acid rain negatively impacts soil quality and the condition of vegetation. In picture 1, one can tell that sample B is not as healthy as samples A and C because of the fact that it had been exposed to excessively acidic water; the fescue grass became yellow and dry, and was comparatively sparse. On the other hand, the fescue grass exposed to sample A became bright green and moist, and it was comparatively dense. Lastly, the grass exposed to sample C became light green and dry, and slightly sparse compared to the other samples. This made it clear that a higher concentration of hydrogen ions in the sample would lower the soils capacity to neutralize the excessive acidity. In addition, these results indicate that a relationship exists between the acidity of the water sample and growth of the fescue grass. Firstly, the condition of the soil and fescue grass in sample A indicates normal, healthy plant growth. Secondly, the condition of the soil and fescue grass in sample B indicates
severely hindered, unhealthy plant growth. Finally, the condition of the soil and fescue grass in sample C indicates moderately hindered, unhealthy plant growth.
Conclusions:
The hypothesis -- "If fescue plants are exposed to substances with different levels of acidity -- one with a pH level of 5, the other with a pH level of 2.4, and the last with a pH level of 4 --, then the grass exposed to the sample with a pH level of 5 (water only) will experience the most growth and be the healthiest, the grass exposed to the vinegar-water mixture with a pH level of 4 will experience less growth and be less healthy, and the grass exposed to the sample of water with a pH level of 2.4 (vinegar only) will undergo the least growth and be the least healthy of the three fescue plants" -- was substantiated by the results of this experiment. There may have been errors in this experiment, but they were not significant enough to compromise the validity of the hypothesis. The results of the experiment made it apparent that an inverse correlation exists between the acidity of water to which the grass is exposed and vegetative growth; therefore, lowering the pH of the water sample hinders vegetative growth even more. "This is particularly true in the northeastern United States, where the rain tends to be most acidic, and often the soil has less capacity to neutralize the acidity"(Acid Rain, EPA). For this reason, the fescue grass exposed to sample A experienced stable, healthy growth, while the fescue grass exposed to sample B experienced hindered, unhealthy growth.
According to the Environmental Protection Agency, the average pH level of acid rain is 4.0. Therefore, sample C -- which was exposed to water with a pH level of 4 -- was the most accurate simulations of vegetation exposed to acid rain. This sample experienced moderately hindered, unhealthy growth. According to the EPA, this is attributes this to the fact that acidic water dissolves and washes away essential base nutrients found in soil -- calcium, magnesium, and potassium. These nutrients are essential to plant growth not only because they are necessary for plants to carry out their life processes normally, but they are also used to neutralize acids and, in a process called "buffering," help soil resist the effects of acid rain. Thus, thin soils are more vulnerable to the pernicious effects of acid rain; the nutrients found in these soils can be more easily eroded and/or dissolved. "Over a long period of time, soils affected by acid rain can become so acidic that aluminum, which occurs naturally in the soil, dissolves and is carried by the rainwater into lakes and streams"(Jack, Gina). Dissolved aluminum is so toxic to aquatic life because it is unlike the insoluble nontoxic form of aluminum hydroxide normally found in soils. Another real-world concern induced by this anthropogenic phenomenon is the fact that acid rain deposits nitrates that can lead to increases in nitrogen in forests. This can lead to nitrogen saturation, which further diminishes the amount of alkaline chemicals -- the essential base nutrients --, and may cause eutrophication in areas where rivers enter the ocean.
As the list of environmental repercussions of acid rain proves, human activities play a large role in the overall health of ecosystems. Most of the SO2 and NOx -- gases that create acid rain -- found in the atmosphere is a result of human activities. Electric power generation produces a majority of these pollutants through fossil fuel combustion, especially coal. In addition, the exhaust from cars releases NOx and SO2 into the air. It is of utmost importance to consider the fact that acid rain causes damage to an ecosystem at a variety of interconnected, ecological levels; hindering plant growth is just one example. Acid rain has other pernicious effects, such as killing fish populations by reducing the alkalinity (buffering capacity) of the water and increasing its concentration of toxic substances such as aluminum; negatively affecting soil health by leaching away important nutrients and minerals, as well as reducing its ability to support healthy plant life; and lowering water quality by increasing the deposition of nitrates that can lead to eutrophication. Some farmers have responded to the unhealthy plant growth caused by acid rain by applying more fertilizers to the soil to compensate for the loss of nutrients, but this is not a sensible solution in the context of the larger environmental issues faced by the world today. "Many governments are now requiring energy producers to clean smoke stacks by using scrubbers which trap pollutants before they are released into the atmosphere and catalytic converters in cars to reduce their emissions. Additionally, alternative energy sources are gaining more prominence today and funding is being given to the restoration of ecosystems damaged by acid rain worldwide" (Briney, Amanda). The passage above provides an example of the stringent environmental policies that need to be implemented in the U.S. -- as well as other parts of the world affected by acid rain -- in order to preclude further deterioration of our ambient environment, as well as avoid one of the many threats to the Earth's delicate ecological balance.
Citations:
"Acid Rain." EPA. Environmental Protection Agency, 10 Sept. 2014. Web. 15 Nov. 2014. <http://www.epa.gov/acidrain/>.
Briney, Amanda. "How Much Should You Worry About Acid Rain?" About. N.p., n.d. Web. 11 Nov. 2014. <http://geography.about.com/od/globalproblemsandissues/a/acidrain.htm>.
Jack, Gina. "What Are the Effects of Acid Rain." New York State Conservationist. Department of Environmental Conservation, Spring 2013. Web. 04 Dec. 2014. <http://www.dec.ny.gov/education/92674.html>.
Norris, Kara. Acid Rain Lab. N.p.: Web. 16 Nov. 2014.
<https://docs.google.com/document/d/1n022jVTyn5F5WnTOl_dbv8JWmLE7h2ety0F9Kmk4_Zg/edit>.
The experiment began with an attempt to make all three of the fescue plants indistinguishable. They were cut down to a length of 6 cm, and had been watered the same amount prior to this experiment. Picture 1 shows the condition of the three samples of grass (samples A, B, and C) before they were exposed to their corresponding samples of water once a week for a total of three weeks. Then, the experiment was conducted, and the data collected. After collecting the aforementioned data, it became apparent that acid rain has pernicious effects on plant growth and soil health; this was further substantiated by the fact that soil exposed to the water with a pH level of 5 (sample A) sustained an aesthetic appearance -- moist and clumped --, while the soil exposed to the water with a pH level of 2.4 (sample B) became dry and unclumped. Sample B resulted in decreased plant growth, and it expedited the falling apart of the soil particles. The fescue plant exposed to the water with a pH level of 4 (sample C) had characteristics similar to sample B in terms of the conditions of the soil and plant growth, which would indicate that an inverse correlation exists between the acidity of water to which the grass is exposed and vegetative growth.
A cursory glance at the three fescue plants would testify to the fact that the acid rain negatively impacts soil quality and the condition of vegetation. In picture 1, one can tell that sample B is not as healthy as samples A and C because of the fact that it had been exposed to excessively acidic water; the fescue grass became yellow and dry, and was comparatively sparse. On the other hand, the fescue grass exposed to sample A became bright green and moist, and it was comparatively dense. Lastly, the grass exposed to sample C became light green and dry, and slightly sparse compared to the other samples. This made it clear that a higher concentration of hydrogen ions in the sample would lower the soils capacity to neutralize the excessive acidity. In addition, these results indicate that a relationship exists between the acidity of the water sample and growth of the fescue grass. Firstly, the condition of the soil and fescue grass in sample A indicates normal, healthy plant growth. Secondly, the condition of the soil and fescue grass in sample B indicates
severely hindered, unhealthy plant growth. Finally, the condition of the soil and fescue grass in sample C indicates moderately hindered, unhealthy plant growth.
Conclusions:
The hypothesis -- "If fescue plants are exposed to substances with different levels of acidity -- one with a pH level of 5, the other with a pH level of 2.4, and the last with a pH level of 4 --, then the grass exposed to the sample with a pH level of 5 (water only) will experience the most growth and be the healthiest, the grass exposed to the vinegar-water mixture with a pH level of 4 will experience less growth and be less healthy, and the grass exposed to the sample of water with a pH level of 2.4 (vinegar only) will undergo the least growth and be the least healthy of the three fescue plants" -- was substantiated by the results of this experiment. There may have been errors in this experiment, but they were not significant enough to compromise the validity of the hypothesis. The results of the experiment made it apparent that an inverse correlation exists between the acidity of water to which the grass is exposed and vegetative growth; therefore, lowering the pH of the water sample hinders vegetative growth even more. "This is particularly true in the northeastern United States, where the rain tends to be most acidic, and often the soil has less capacity to neutralize the acidity"(Acid Rain, EPA). For this reason, the fescue grass exposed to sample A experienced stable, healthy growth, while the fescue grass exposed to sample B experienced hindered, unhealthy growth.
According to the Environmental Protection Agency, the average pH level of acid rain is 4.0. Therefore, sample C -- which was exposed to water with a pH level of 4 -- was the most accurate simulations of vegetation exposed to acid rain. This sample experienced moderately hindered, unhealthy growth. According to the EPA, this is attributes this to the fact that acidic water dissolves and washes away essential base nutrients found in soil -- calcium, magnesium, and potassium. These nutrients are essential to plant growth not only because they are necessary for plants to carry out their life processes normally, but they are also used to neutralize acids and, in a process called "buffering," help soil resist the effects of acid rain. Thus, thin soils are more vulnerable to the pernicious effects of acid rain; the nutrients found in these soils can be more easily eroded and/or dissolved. "Over a long period of time, soils affected by acid rain can become so acidic that aluminum, which occurs naturally in the soil, dissolves and is carried by the rainwater into lakes and streams"(Jack, Gina). Dissolved aluminum is so toxic to aquatic life because it is unlike the insoluble nontoxic form of aluminum hydroxide normally found in soils. Another real-world concern induced by this anthropogenic phenomenon is the fact that acid rain deposits nitrates that can lead to increases in nitrogen in forests. This can lead to nitrogen saturation, which further diminishes the amount of alkaline chemicals -- the essential base nutrients --, and may cause eutrophication in areas where rivers enter the ocean.
As the list of environmental repercussions of acid rain proves, human activities play a large role in the overall health of ecosystems. Most of the SO2 and NOx -- gases that create acid rain -- found in the atmosphere is a result of human activities. Electric power generation produces a majority of these pollutants through fossil fuel combustion, especially coal. In addition, the exhaust from cars releases NOx and SO2 into the air. It is of utmost importance to consider the fact that acid rain causes damage to an ecosystem at a variety of interconnected, ecological levels; hindering plant growth is just one example. Acid rain has other pernicious effects, such as killing fish populations by reducing the alkalinity (buffering capacity) of the water and increasing its concentration of toxic substances such as aluminum; negatively affecting soil health by leaching away important nutrients and minerals, as well as reducing its ability to support healthy plant life; and lowering water quality by increasing the deposition of nitrates that can lead to eutrophication. Some farmers have responded to the unhealthy plant growth caused by acid rain by applying more fertilizers to the soil to compensate for the loss of nutrients, but this is not a sensible solution in the context of the larger environmental issues faced by the world today. "Many governments are now requiring energy producers to clean smoke stacks by using scrubbers which trap pollutants before they are released into the atmosphere and catalytic converters in cars to reduce their emissions. Additionally, alternative energy sources are gaining more prominence today and funding is being given to the restoration of ecosystems damaged by acid rain worldwide" (Briney, Amanda). The passage above provides an example of the stringent environmental policies that need to be implemented in the U.S. -- as well as other parts of the world affected by acid rain -- in order to preclude further deterioration of our ambient environment, as well as avoid one of the many threats to the Earth's delicate ecological balance.
Citations:
"Acid Rain." EPA. Environmental Protection Agency, 10 Sept. 2014. Web. 15 Nov. 2014. <http://www.epa.gov/acidrain/>.
Briney, Amanda. "How Much Should You Worry About Acid Rain?" About. N.p., n.d. Web. 11 Nov. 2014. <http://geography.about.com/od/globalproblemsandissues/a/acidrain.htm>.
Jack, Gina. "What Are the Effects of Acid Rain." New York State Conservationist. Department of Environmental Conservation, Spring 2013. Web. 04 Dec. 2014. <http://www.dec.ny.gov/education/92674.html>.
Norris, Kara. Acid Rain Lab. N.p.: Web. 16 Nov. 2014.
<https://docs.google.com/document/d/1n022jVTyn5F5WnTOl_dbv8JWmLE7h2ety0F9Kmk4_Zg/edit>.