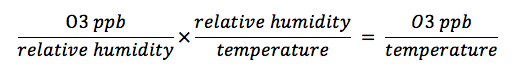Collaborators:
David Delgado, Hailey Hendriks, Caroline Knowles, Hannah Midles and Rachel Werner
David Delgado, Hailey Hendriks, Caroline Knowles, Hannah Midles and Rachel Werner
Pre-lab Questions:
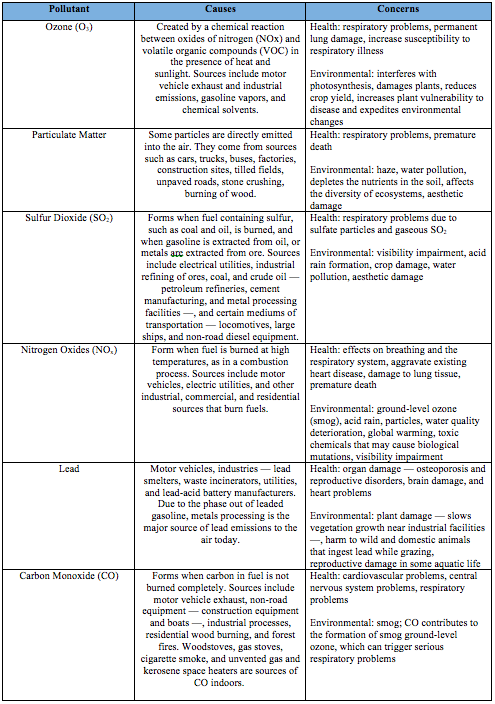
1. Create a chart showing the six air pollutants, causes, and concerns from the information in the pre-lab.
1. Create a chart showing the six air pollutants, causes, and concerns from the information in the pre-lab.
2. Why is ground level ozone highest in the summer and almost nonexistent in the winter? How might this affect our results as we test the air during this part of the year?
Ground level ozone is highest in the summer and almost nonexistent in the winter because sunlight and hot weather are what cause it to develop in harmful concentrations. This is due to the chemical reaction that produces ozone: VOC + NOx + Heat + Sunlight --> Ozone. In the summer, there is more heat and sunlight, which provide the energy needed for the chemical reaction to occur, thereby increasing the concentration of ground level ozone. This might cause the test results to be lower than what they would be in the summer, seeing as though we are experiencing a pernicious drop in local temperatures throughout the southern U.S.
3. Many third world countries still use leaded gasoline (since they have older cars or since they have not forced changes). What concerns may the governments of these countries need to deal with?
Lead is a pollutant that causes both health and environmental problems. The governments of these third world countries would have to deal with their citizens experiencing organ damage -- osteoporosis and reproductive disorders, brain damage, and heart problems. Some of the environmental concerns that they would face include plant damage -- slows vegetation growth near industrial facilities --, harm to wild and domestic animals that ingest lead while grazing, and reproductive damage in some aquatic life.
4. Use the Internet to find what parts of North Carolina are not in compliance with the Standards of the Clean Air Act. You may give either general areas or specific counties.
The highest rating the EPA will allow for a community for ground level ozone is 80 ppb. Any community with over 80 ppb for an average over three years is in noncompliance with the Clean Air Act. Based on this information, I would predict that Wake Forest is leaning toward noncompliance due to the growing amount of urbanization occurring in this town. Ozone levels increase as more motor vehicle exhaust, industrial emissions, and chemical solvents are added to the atmosphere. According to the Environmental Protection Agency, as of July 2, 2014, the counties of North Carolina that are not in compliance with the standards of the Clean Air Act are Cabarrus County, Gaston County, Iredell County, Lincoln County, Mecklenburg County, Rowan County, and Union County.
Introduction & Problem:
Criteria Air Pollutants -- as defined by the Environmental Protection Agency (EPA) -- serve as crucial indicators of regional air quality. However, this lab will only focus on two of the six aforementioned pollutants -- ground-level ozone (O3) and particulate matter (PM) --, as they can be readily assessed without the use of expensive models that correspond to the actual meteorological conditions. Certain aspects of the environment have been downplayed to study and analyze these observation-derived relationships. Nevertheless, the effects of these air pollutants cannot be understated. Ozone -- created by chemical reactions between oxides of nitrogen (NOx) and volatile organic compounds (VOCs) in the presence of sunlight -- and particulate matter -- a combination of solid particles and liquid droplets in the air -- are increasing in concentration, and it is a pervasive problem in North America. Although they are naturally occurring substances (i.e. stratospheric ozone and particulate matter released from sources such as windblown dust and wildfires), anthropogenic activity has been shown to aggravate the problem by releasing excess O3 and PM into the atmosphere. "Motor vehicle exhaust and industrial emissions, gasoline vapors, and chemical solvents are some of the major sources of NOx and VOCs that help to form ozone"(Norris, Kara). Likewise, particulate matter can be directly emitted into the air by sources such as cars, trucks, buses, factories, construction sites, burning of wood and tilled fields.
The ecological and health repercussions of these ambient air pollutants are just as complex in nature as the factors influencing their formation. According to the EPA, breathing O3 can exacerbate respiratory problems, and it has been shown to have pernicious effects on sensitive vegetation and ecosystems. In addition, PM has been linked to a range of serious respiratory and cardiovascular health problems, along with the concerns associated with atmospheric deposition -- making lakes acidic, depleting the nutrients in soil, damaging sensitive forests and farm crops, and affecting the diversity of ecosystems. Therefore, it is important to understand the variables that affect the formation of ground level ozone and particulate matter, and then use this information to develop pragmatic solutions to the problem. For example, it is interesting to note that ground-level ozone is more prevalent in the summer; it is known as "summertime air pollutant" due to the fact that "sunlight and hot weather cause ground-level ozone to form in harmful concentrations in the air"(Norris, Kara). In addition, particulate matter concentrations may increase with the presence of winds that carry particulate matter from its source and deposits it in a new location.
To summarize the conclusions drawn from these studies, it becomes necessary to address the question, "How does the indoor air quality of Heritage High School compare to the outdoor air quality?" Although this question appears to oversimplify the topic at hand, it actually does the complete opposite: Answering the aforementioned question would necessitate a comprehensive understanding of the meteorological and chemical regimes from which an assessment of the ambient air quality is derived. From there, the assessment of the outdoor air quality can be compared with similar studies disclosed by the EPA. For instance, as of July 2, 2014, the counties of North Carolina that are not in compliance with the standards of the Clean Air Act are Cabarrus County, Gaston County, Iredell County, Lincoln County, Mecklenburg County, Rowan County, and Union County. Although Wake Forest may be in compliance with the standards of the Clean Air Act, the problem should not be overlooked -- especially as the town continues to experience urban development. The effects of rising levels of O3 and PM in the atmosphere are not limited to health and environmental concerns, but it also has significant economic implications that arise from the deleterious effects of air pollutants such as ozone and particulate matter.
Hypothesis:
If the air quality inside and outside of Heritage High School -- in terms of Ozone and PM measurements -- is measured, then the air quality outside will be lower than inside because there is more exposure to air pollutants outside while air filters found inside clear up particulate matter and ozone to hamper the infiltration of outdoor ozone and particulate matter.
Parts of the Experiment:
Materials & Methods:
a) Materials:
b) Method:
A. Preparation
Ground level ozone is highest in the summer and almost nonexistent in the winter because sunlight and hot weather are what cause it to develop in harmful concentrations. This is due to the chemical reaction that produces ozone: VOC + NOx + Heat + Sunlight --> Ozone. In the summer, there is more heat and sunlight, which provide the energy needed for the chemical reaction to occur, thereby increasing the concentration of ground level ozone. This might cause the test results to be lower than what they would be in the summer, seeing as though we are experiencing a pernicious drop in local temperatures throughout the southern U.S.
3. Many third world countries still use leaded gasoline (since they have older cars or since they have not forced changes). What concerns may the governments of these countries need to deal with?
Lead is a pollutant that causes both health and environmental problems. The governments of these third world countries would have to deal with their citizens experiencing organ damage -- osteoporosis and reproductive disorders, brain damage, and heart problems. Some of the environmental concerns that they would face include plant damage -- slows vegetation growth near industrial facilities --, harm to wild and domestic animals that ingest lead while grazing, and reproductive damage in some aquatic life.
4. Use the Internet to find what parts of North Carolina are not in compliance with the Standards of the Clean Air Act. You may give either general areas or specific counties.
The highest rating the EPA will allow for a community for ground level ozone is 80 ppb. Any community with over 80 ppb for an average over three years is in noncompliance with the Clean Air Act. Based on this information, I would predict that Wake Forest is leaning toward noncompliance due to the growing amount of urbanization occurring in this town. Ozone levels increase as more motor vehicle exhaust, industrial emissions, and chemical solvents are added to the atmosphere. According to the Environmental Protection Agency, as of July 2, 2014, the counties of North Carolina that are not in compliance with the standards of the Clean Air Act are Cabarrus County, Gaston County, Iredell County, Lincoln County, Mecklenburg County, Rowan County, and Union County.
Introduction & Problem:
Criteria Air Pollutants -- as defined by the Environmental Protection Agency (EPA) -- serve as crucial indicators of regional air quality. However, this lab will only focus on two of the six aforementioned pollutants -- ground-level ozone (O3) and particulate matter (PM) --, as they can be readily assessed without the use of expensive models that correspond to the actual meteorological conditions. Certain aspects of the environment have been downplayed to study and analyze these observation-derived relationships. Nevertheless, the effects of these air pollutants cannot be understated. Ozone -- created by chemical reactions between oxides of nitrogen (NOx) and volatile organic compounds (VOCs) in the presence of sunlight -- and particulate matter -- a combination of solid particles and liquid droplets in the air -- are increasing in concentration, and it is a pervasive problem in North America. Although they are naturally occurring substances (i.e. stratospheric ozone and particulate matter released from sources such as windblown dust and wildfires), anthropogenic activity has been shown to aggravate the problem by releasing excess O3 and PM into the atmosphere. "Motor vehicle exhaust and industrial emissions, gasoline vapors, and chemical solvents are some of the major sources of NOx and VOCs that help to form ozone"(Norris, Kara). Likewise, particulate matter can be directly emitted into the air by sources such as cars, trucks, buses, factories, construction sites, burning of wood and tilled fields.
The ecological and health repercussions of these ambient air pollutants are just as complex in nature as the factors influencing their formation. According to the EPA, breathing O3 can exacerbate respiratory problems, and it has been shown to have pernicious effects on sensitive vegetation and ecosystems. In addition, PM has been linked to a range of serious respiratory and cardiovascular health problems, along with the concerns associated with atmospheric deposition -- making lakes acidic, depleting the nutrients in soil, damaging sensitive forests and farm crops, and affecting the diversity of ecosystems. Therefore, it is important to understand the variables that affect the formation of ground level ozone and particulate matter, and then use this information to develop pragmatic solutions to the problem. For example, it is interesting to note that ground-level ozone is more prevalent in the summer; it is known as "summertime air pollutant" due to the fact that "sunlight and hot weather cause ground-level ozone to form in harmful concentrations in the air"(Norris, Kara). In addition, particulate matter concentrations may increase with the presence of winds that carry particulate matter from its source and deposits it in a new location.
To summarize the conclusions drawn from these studies, it becomes necessary to address the question, "How does the indoor air quality of Heritage High School compare to the outdoor air quality?" Although this question appears to oversimplify the topic at hand, it actually does the complete opposite: Answering the aforementioned question would necessitate a comprehensive understanding of the meteorological and chemical regimes from which an assessment of the ambient air quality is derived. From there, the assessment of the outdoor air quality can be compared with similar studies disclosed by the EPA. For instance, as of July 2, 2014, the counties of North Carolina that are not in compliance with the standards of the Clean Air Act are Cabarrus County, Gaston County, Iredell County, Lincoln County, Mecklenburg County, Rowan County, and Union County. Although Wake Forest may be in compliance with the standards of the Clean Air Act, the problem should not be overlooked -- especially as the town continues to experience urban development. The effects of rising levels of O3 and PM in the atmosphere are not limited to health and environmental concerns, but it also has significant economic implications that arise from the deleterious effects of air pollutants such as ozone and particulate matter.
Hypothesis:
If the air quality inside and outside of Heritage High School -- in terms of Ozone and PM measurements -- is measured, then the air quality outside will be lower than inside because there is more exposure to air pollutants outside while air filters found inside clear up particulate matter and ozone to hamper the infiltration of outdoor ozone and particulate matter.
Parts of the Experiment:
- This experiment does not have a control group.
- The experimental group is constituted by the ozone monitoring strips and the particulate matter collectors that are placed inside and outside of Heritage High School.
- The independent variable is the location in which the ozone monitoring strips and the particulate matter collectors are placed.
- The dependent variable is the air quality measured inside and outside of Heritage High School, in terms of the ground level ozone concentration and the particulate matter concentration of these different locations.
- The controlled variables include the materials and the time period in which the ozone and particulate matter measurements are recorded.
Materials & Methods:
a) Materials:
- Potassium Iodide
- Corn starch
- Schoenbein paper
- Schoenbein Number chart
- Relative Humidity Schoenbein Number chart
- Beakers
- Spatulas
- Large Graduated Cylinder
- Test Tube Rack
- Filter Paper
- Microscope slides
- Petroleum Jelly (vaseline)
- String
- Q-tips
- Microscopes
b) Method:
A. Preparation
- Half the class should work on preparing the ozone monitoring test strips by placing 100ml of water in a 250 ml beaker, and adding 5g of cornstarch. Then heat and stir the mixture until it gels (when the mixture becomes thick and clear). Remove the solution from heat, add 1 g of potassium iodide, stir well, and allow to cool. After the solution has cooled, lay a piece of filter paper on a petri and brush the paste onto the filter paper. Turn the filter paper and brush the paste on the other side. Hang up and allow the filter paper to dry. Cut the papers into 1 inch-wide strips. Store the strips in a plastic bag out of sunlight.
- The other half of the class will prepare Particulate matter collectors by smearing a thin layer of petroleum jelly onto a microscope slides. Try to make the layer as even as possible. Use a q-tip to lightly smear the specimen side of each slide. You will need to prepare 2 slides per group and place them in a slide container to avoid smearing the petroleum jelly on anyone.
- Each group will place one labeled slide on the exterior of the school. Some groups should choose areas that would receive few particulates, others should choose locations that produce more particulates. (Keep in mind sources of particulate matter)
- One member from each group should take the second slide and place it in a location inside the school.
- You should then place your ozone monitoring strip outside for 8 hrs. Before placing the strip outside, dip the strip in distilled water, then attach the strip out of sunlight. When placing your strip find the relative humidity for that day.
- The next day, each group should collect their slide and ozone monitoring strip.
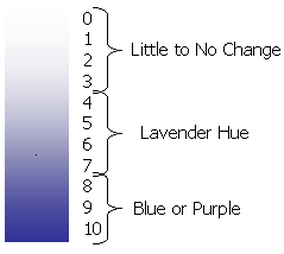
- Dunk each strip into distilled water. Use the chart at the bottom left to determine the Schoenbein Number of your strip.
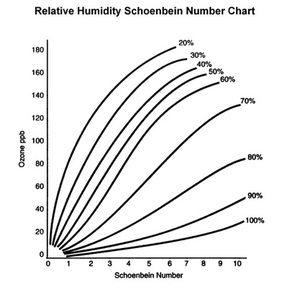
6. Use the relative humidity and the Schoenbein number to calculate the concentration of ground level ozone using the chart above (Relative Humidity Schoenbein Number Chart).
7. Look at your particulate matter slide under the microscope. First decide the amount of particulates and then attempt to estimate the source of the PM. Your group will need to decide how you can classify what type of PM was collected.
8. Record all of the lab information in your notebook.
Data & Data Analysis:
a) Data:
7. Look at your particulate matter slide under the microscope. First decide the amount of particulates and then attempt to estimate the source of the PM. Your group will need to decide how you can classify what type of PM was collected.
8. Record all of the lab information in your notebook.
Data & Data Analysis:
a) Data:
|
According to the EPA, the ideal levels of humidity for your living space will be less than 60% in the summer and between 25 — 40% in the winter. Humid air
contains more heat than dry air at the same temperature, which catalyzes the formation of ground-level ozone according to the formula: VOC + NOx + Heat + Sunlight --> O3 (Ozone). |
Data Tables:
Table 1: Group Ozone and PM Results
Table 2: Class Ozone and PM Results
|
b) Data Analysis:
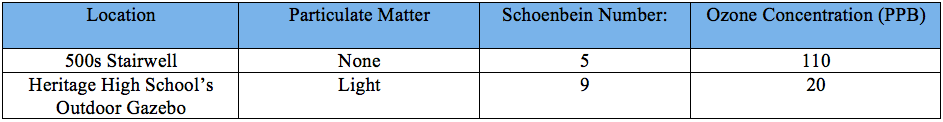
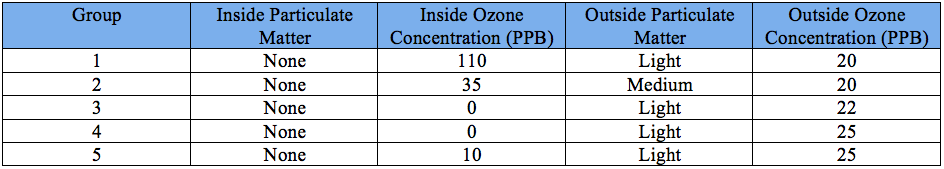
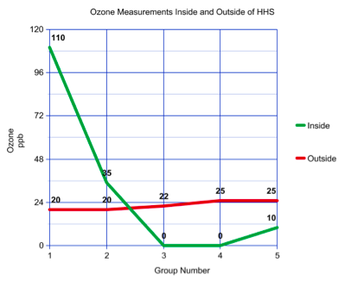
The experiment began with the application of a mixture on the Schoenbein paper -- which would be a test for ozone -- and smearing petroleum jelly on notecards that would be used to measure the ambient particulate matter. Then, the focus of the group became the idyllic allocation of the ozone monitoring strips and the particulate matter collectors inside and outside of Heritage High School. As shown in the pictures above, one of each of the PM and O3 indicators was placed inside, while the other was placed outside. After recording the data, it became apparent that the formation of ground-level ozone was inextricably linked to the relative humidity both indoors and outdoors. It appears that relative humidity and temperature are inversely related. Specifically, if one takes that temperature and raises it while keeping the same amount of water vapor -- the maximum amount of water that can be held becomes much higher -- then the relative humidity will be much lower. Thus, the higher temperature inside resulted in a comparatively lower relative humidity, which in turn led to a higher concentration of ozone in parts per billion (ppb) being recorded by groups 1 and 2 as compared to the outside ozone concentration. However, the discrepancies in ozone measurements indoors makes this observation questionable. As shown in the graph above, the ozone concentration recorded indoors varied significantly from one group to the other, which may indicate that indoor air pollution is just as volatile as outdoor air pollution. Nevertheless, the outdoor ozone measurements were quite homogenous, with very little variation between groups. The mean outdoor ozone concentration recorded by groups was 22.4 ppb.
The other air pollutant assessed in this experiment -- particulate matter (PM) -- also produced intriguing results. By examining the particulate matter slides under a microscope and using the charts provided in the lab, the relative particulate matter concentration of each location was successfully determined: Heritage High School's outdoor gazebo - light, 500-600s stairwell - none. The lowest concentration of PM was indoors for all groups, while the outdoor concentration had the highest. Although the results were quite homogenous, the PM collected could not be classified at first. It was later determined that the particulates captured had been mostly coarse particles, such as dusts and liquid droplets that were blown by the wind. However, the ambient particulate matter did not show high concentrations of fine particles -- particulates that are known for their deleterious effects on human health. On the other hand, the class reached a consensus on the PM collected inside. The fact that no PM was found inside of Heritage High School -- both coarse and fine particles -- is most likely due to the lack of a emission source indoors and/or the fact that the building is sealed and ventilated well, which hinders the amount of particulates entering the building from outside.
Conclusions:
The results of this experiment do not entirely support the hypothesis: "If the air quality inside and outside of Heritage High School -- in terms of Ozone and PM measurements -- is measured, then the air quality outside will be lower than inside because there is more exposure to air pollutants outside while air filters found inside clear up particulate matter and ozone to hamper the infiltration of outdoor ozone and particulate matter." Some groups -- groups 1 and 2 -- found that the indoor ozone concentration was significantly higher than the outdoor ozone concentration. Although this incurred much doubt in the accuracy of the measurements of the air quality, further analysis provided an explanation for this phenomenon. Firstly, it is known that ground-level ozone is highest in the summer and almost nonexistent in the winter because sunlight and hot weather are what cause it to develop in harmful concentrations. Therefore, low concentrations of ambient ozone were recorded by every group, allowing Wake Forest to meet the standards of the Clean Air Act on this day. Secondly, all of the ozone strips placed outside could be found around the gazebo; this location limited the exposure to the motor vehicle exhaust -- a source of nitrogen oxide (NOx) -- coming from the student parking lot. Thus, with a shortage of reactants, the air quality was found to be well above the standard imposed by the EPA of 80 ppb. In addition, the relative humidity used by the class to estimate the ozone concentration was the measurement for the day that the data was recorded, and not an average of the relative humidity during the span of the experiment. Lastly, there are several ways that ozone can enter the school. For example, some air cleaners, which are intended to control indoor air pollution and remove causing chemicals, actually produce ozone -- "there is evidence to show that at concentrations that do not exceed public health standards, ozone is not effective at removing many odor-causing chemicals"(Environmental Protection Agency, 2014). Some cleaning products and building materials also contain elevated amounts of VOCs, and with the limited amount of ventilation inside the school these pollutants can aggregate quickly. Nevertheless,
the ground level ozone concentration of a city on a single day cannot be generalized to describe the normal air quality of that city for an extended period of time, for there are many variables that affect ground ozone level concentrations. In order to achieve accurate measurements of the air quality of a city, one must calculate the ground level ozone of different seasons, as well as areas.
Particulate matter (PM) was the other ambient air pollutant that was recorded in this experiment. Unlike the ozone measurements, PM produced conclusive results that support the hypothesis in its entirety. None of the groups found particulates to be present indoors, while most found that the outdoor concentration of particulate matter to be light. Perhaps the most important observation that can be deduced from an assessment of the PM levels is not the amount, but rather the type of PM that was collected along with its source.
"What are the size-distribution, chemical-composition, and mass-emission rates of PM emitted from primary-particle sources in the U.S, and what are the emissions of the reactive gases that lead to secondary particle formation through atmospheric chemical reactions?"("Particulate Matter," EPA). According to the EPA, size-distribution is one way to classify the type of PM collected: "PM can be principally characterized as discrete particles spanning several orders of magnitude in size"(Environmental Protection Agency, 2014). This method of classification is particularly important in terms of the attention that PM has received from governments in recent years. Fine particles -- often referred to as PM 2.5 -- are directly emitted from combustion sources -- coal, oil, diesel, gasoline, wood, as well as high temperature process sources such as smelters and steel mills -- and are also formed secondarily from gaseous precursors such as sulfur dioxide, nitrogen oxides, or organic compounds. With this information in mind, it was not expected for the PM samples to reach malignant concentrations, especially since there is a low concentration of combustion sources nearby and the low concentration of ozone would indicate that there are low levels of certain chemicals (i.e. NOx and VOCs) needed to create fine particles through atmospheric chemical reactions. This warrants the conclusion that the few PM that was collected was could most likely be classified as coarse particles, or particles dominated by resuspended dusts from construction sites and stone crushing. As research expands, more people become cognizant of the pernicious effects of fine particles on human health -- such as aggravating of respiratory and cardiovascular disease. This has allowed PM to be added to the requirements stipulated by the Clean Air Act.
Ozone formation is influenced by a plethora of factors, including temperature, humidity, winds, and the presence of other chemicals in the atmosphere. On top of that, one must take into consideration the effects of climate change on ozone and other atmospheric conditions. Nevertheless, ground-level ozone is a pernicious air pollutant that is known to aggravate respiratory problems and environmental concerns. For this reason, groups did their best to obtain accurate results of the air quality in Wake Forest. However, the data proves that these efforts may have been thwarted by possible errors in the experiment, as well as complex meteorological conditions that influenced ground-level ozone formation. One of these factors is described below:
The warmer the air is in the atmosphere, the higher the ambient ozone concentration. This conclusion points to an important proportional relationship that should be considered in this experiment – the relationship between relative humidity and temperature. When the actual amount of water vapor in the air remains constant, an inverse relationship can exist between relative humidity and temperature. The dew point temperatures are typically closer to the current temperature in cold air, making the relative humidity, or the percent of water vapor in the air from what the water can hold, much higher. But if one takes that temperature and raises it while keeping the same amount of water vapor -- the maximum amount of water that can be held becomes much higher -- then the relative humidity will be much lower. Similarly, an inverse relationship exists between relative humidity and the ozone concentration in parts per billion (ppb), as indicated by the “Relative Humidity Schoenbein Number Chart.” As the formula below demonstrates, these relationships can be used to describe one global phenomenon that has become very controversial. Ozone concentrations in the stratosphere have been shown to decrease as temperatures rise due to global warming. Climate change is in turn expedited by the depletion of ozone in the stratosphere, creating a positive feedback loop associated with changes in temperature: Decreased ozone in the stratosphere results in lower temperatures. “The more ozone destruction in the stratosphere, the colder it would get just because there was less ozone. And the colder it would get, the more ozone depletion would occur”(Allen, Jeannie).
The experiment began with the application of a mixture on the Schoenbein paper -- which would be a test for ozone -- and smearing petroleum jelly on notecards that would be used to measure the ambient particulate matter. Then, the focus of the group became the idyllic allocation of the ozone monitoring strips and the particulate matter collectors inside and outside of Heritage High School. As shown in the pictures above, one of each of the PM and O3 indicators was placed inside, while the other was placed outside. After recording the data, it became apparent that the formation of ground-level ozone was inextricably linked to the relative humidity both indoors and outdoors. It appears that relative humidity and temperature are inversely related. Specifically, if one takes that temperature and raises it while keeping the same amount of water vapor -- the maximum amount of water that can be held becomes much higher -- then the relative humidity will be much lower. Thus, the higher temperature inside resulted in a comparatively lower relative humidity, which in turn led to a higher concentration of ozone in parts per billion (ppb) being recorded by groups 1 and 2 as compared to the outside ozone concentration. However, the discrepancies in ozone measurements indoors makes this observation questionable. As shown in the graph above, the ozone concentration recorded indoors varied significantly from one group to the other, which may indicate that indoor air pollution is just as volatile as outdoor air pollution. Nevertheless, the outdoor ozone measurements were quite homogenous, with very little variation between groups. The mean outdoor ozone concentration recorded by groups was 22.4 ppb.
The other air pollutant assessed in this experiment -- particulate matter (PM) -- also produced intriguing results. By examining the particulate matter slides under a microscope and using the charts provided in the lab, the relative particulate matter concentration of each location was successfully determined: Heritage High School's outdoor gazebo - light, 500-600s stairwell - none. The lowest concentration of PM was indoors for all groups, while the outdoor concentration had the highest. Although the results were quite homogenous, the PM collected could not be classified at first. It was later determined that the particulates captured had been mostly coarse particles, such as dusts and liquid droplets that were blown by the wind. However, the ambient particulate matter did not show high concentrations of fine particles -- particulates that are known for their deleterious effects on human health. On the other hand, the class reached a consensus on the PM collected inside. The fact that no PM was found inside of Heritage High School -- both coarse and fine particles -- is most likely due to the lack of a emission source indoors and/or the fact that the building is sealed and ventilated well, which hinders the amount of particulates entering the building from outside.
Conclusions:
The results of this experiment do not entirely support the hypothesis: "If the air quality inside and outside of Heritage High School -- in terms of Ozone and PM measurements -- is measured, then the air quality outside will be lower than inside because there is more exposure to air pollutants outside while air filters found inside clear up particulate matter and ozone to hamper the infiltration of outdoor ozone and particulate matter." Some groups -- groups 1 and 2 -- found that the indoor ozone concentration was significantly higher than the outdoor ozone concentration. Although this incurred much doubt in the accuracy of the measurements of the air quality, further analysis provided an explanation for this phenomenon. Firstly, it is known that ground-level ozone is highest in the summer and almost nonexistent in the winter because sunlight and hot weather are what cause it to develop in harmful concentrations. Therefore, low concentrations of ambient ozone were recorded by every group, allowing Wake Forest to meet the standards of the Clean Air Act on this day. Secondly, all of the ozone strips placed outside could be found around the gazebo; this location limited the exposure to the motor vehicle exhaust -- a source of nitrogen oxide (NOx) -- coming from the student parking lot. Thus, with a shortage of reactants, the air quality was found to be well above the standard imposed by the EPA of 80 ppb. In addition, the relative humidity used by the class to estimate the ozone concentration was the measurement for the day that the data was recorded, and not an average of the relative humidity during the span of the experiment. Lastly, there are several ways that ozone can enter the school. For example, some air cleaners, which are intended to control indoor air pollution and remove causing chemicals, actually produce ozone -- "there is evidence to show that at concentrations that do not exceed public health standards, ozone is not effective at removing many odor-causing chemicals"(Environmental Protection Agency, 2014). Some cleaning products and building materials also contain elevated amounts of VOCs, and with the limited amount of ventilation inside the school these pollutants can aggregate quickly. Nevertheless,
the ground level ozone concentration of a city on a single day cannot be generalized to describe the normal air quality of that city for an extended period of time, for there are many variables that affect ground ozone level concentrations. In order to achieve accurate measurements of the air quality of a city, one must calculate the ground level ozone of different seasons, as well as areas.
Particulate matter (PM) was the other ambient air pollutant that was recorded in this experiment. Unlike the ozone measurements, PM produced conclusive results that support the hypothesis in its entirety. None of the groups found particulates to be present indoors, while most found that the outdoor concentration of particulate matter to be light. Perhaps the most important observation that can be deduced from an assessment of the PM levels is not the amount, but rather the type of PM that was collected along with its source.
"What are the size-distribution, chemical-composition, and mass-emission rates of PM emitted from primary-particle sources in the U.S, and what are the emissions of the reactive gases that lead to secondary particle formation through atmospheric chemical reactions?"("Particulate Matter," EPA). According to the EPA, size-distribution is one way to classify the type of PM collected: "PM can be principally characterized as discrete particles spanning several orders of magnitude in size"(Environmental Protection Agency, 2014). This method of classification is particularly important in terms of the attention that PM has received from governments in recent years. Fine particles -- often referred to as PM 2.5 -- are directly emitted from combustion sources -- coal, oil, diesel, gasoline, wood, as well as high temperature process sources such as smelters and steel mills -- and are also formed secondarily from gaseous precursors such as sulfur dioxide, nitrogen oxides, or organic compounds. With this information in mind, it was not expected for the PM samples to reach malignant concentrations, especially since there is a low concentration of combustion sources nearby and the low concentration of ozone would indicate that there are low levels of certain chemicals (i.e. NOx and VOCs) needed to create fine particles through atmospheric chemical reactions. This warrants the conclusion that the few PM that was collected was could most likely be classified as coarse particles, or particles dominated by resuspended dusts from construction sites and stone crushing. As research expands, more people become cognizant of the pernicious effects of fine particles on human health -- such as aggravating of respiratory and cardiovascular disease. This has allowed PM to be added to the requirements stipulated by the Clean Air Act.
Ozone formation is influenced by a plethora of factors, including temperature, humidity, winds, and the presence of other chemicals in the atmosphere. On top of that, one must take into consideration the effects of climate change on ozone and other atmospheric conditions. Nevertheless, ground-level ozone is a pernicious air pollutant that is known to aggravate respiratory problems and environmental concerns. For this reason, groups did their best to obtain accurate results of the air quality in Wake Forest. However, the data proves that these efforts may have been thwarted by possible errors in the experiment, as well as complex meteorological conditions that influenced ground-level ozone formation. One of these factors is described below:
The warmer the air is in the atmosphere, the higher the ambient ozone concentration. This conclusion points to an important proportional relationship that should be considered in this experiment – the relationship between relative humidity and temperature. When the actual amount of water vapor in the air remains constant, an inverse relationship can exist between relative humidity and temperature. The dew point temperatures are typically closer to the current temperature in cold air, making the relative humidity, or the percent of water vapor in the air from what the water can hold, much higher. But if one takes that temperature and raises it while keeping the same amount of water vapor -- the maximum amount of water that can be held becomes much higher -- then the relative humidity will be much lower. Similarly, an inverse relationship exists between relative humidity and the ozone concentration in parts per billion (ppb), as indicated by the “Relative Humidity Schoenbein Number Chart.” As the formula below demonstrates, these relationships can be used to describe one global phenomenon that has become very controversial. Ozone concentrations in the stratosphere have been shown to decrease as temperatures rise due to global warming. Climate change is in turn expedited by the depletion of ozone in the stratosphere, creating a positive feedback loop associated with changes in temperature: Decreased ozone in the stratosphere results in lower temperatures. “The more ozone destruction in the stratosphere, the colder it would get just because there was less ozone. And the colder it would get, the more ozone depletion would occur”(Allen, Jeannie).
Of course, this is only one of the relationships that have been formulated based on studies of ozone. It would be more accurate to say that temperature, humidity, winds, and the presence of other chemicals in the atmosphere influence ozone formation, and the presence of ozone, in turn, affects those atmospheric constituents. As a result of the complexity of this study, it would be impossible at this point for scientists to thoroughly assess the implications of climate change on air quality or population exposures to high concentrations of ground-level ozone. The limited studies that do exist only manage to suggest that a nonlinear relationship exists between temperature and ozone concentrations at ground level: “Below temperatures of 22-26ºC (70-80ºF), there is no relationship between ozone concentrations and temperature; above 32ºC (90ºF), there is a strong positive relationship”(The Regional Impacts of Climate Change, IPPC). In other words, only high temperatures can be linked to ozone formation at ground level, and it thus becomes a pivotal atmospheric condition for high ozone concentrations. It follows that ozone concentrations at ground level continue to be the most pervasive air pollution problem in North America. A consensus among climate scientists corroborates the fact that ground-level ozone is an anthropogenic phenomenon. This air pollutant is a product of motor vehicle exhaust, chemical solvents, gasoline vapors and industrial emissions -- these sources add to the anthropogenic and natural emissions of NOx, leading to ozone formation if adequately synthesized in the presence of sunlight and heat. For this reason, highly urbanized communities and industrial centers (i.e. in the northeastern and southwestern U.S.) are responsible for inducing high ozone concentrations that do not meet the ozone requirements of the Clean Air Act. Another reason for this has to do with a concept that has already been explained: rising temperatures. Weather and climate have a significant effect on the location of air pollution in the United States, especially ground-level ozone. Concepts learned in AP Environmental Science further support the location of the counties in Non-attainment, such as the fact that prevailing winds carry primary and secondary air pollutants from the West to the East, thereby expediting air pollution in the East.
According to the Environmental Protection Agency, the Clean Air Act has led to economic prosperity in recent years: The United States environmental protection technologies industry, [which includes all environmental media, environmental cleanup, and resource recovery,] by 2007, “was generating approximately $282 billion in revenues, producing $40 billion in exports, and supporting 1.6 million jobs” (Environmental Protection Agency, 2013). In addition, due to the deleterious effects of air pollutants, such as ozone and particulate matter, on public health, a proliferation in air pollution can lead to a significant attrition in the number of workers, which presents negative effects on the economy. In such an unfavorable scenario, air quality monitoring precludes not only harmful effects on the environment, but also the economy. Increased exposure to PM would be harmful to any nation's economy, for there are jobs that need to be carried out. Premature deaths and aggravated respiratory problems disrupt the nation's capitalistic economy by reducing the labor pool in the U.S. and putting people out of work, "as indicated by increased hospital admissions and emergency room visits, school absences, work loss days, and restricted activity days"("Particulate Matter," EPA). Finally, air pollutants, such as sulfur dioxide, can produce acid rain, which kills vegetation, leading to decreased crop yields, and ultimately harming the agricultural industry. From these conclusions, it is important to walk away with one thing in mind: Just as atmospheric conditions and O3 and PM concentrations influence one another, human activities influence the formation of these air pollutants and those pollutants will inevitably affect them.
Citations:
Allen, Jeannie. "National Aeronautics and Space Administration." NASA GISS: Research Features: Ozone and Climate Change. NASA, Feb. 2004. Web. 12 Dec. 2014. <http://www.giss.nasa.gov/research/features/200402_tango/>.
Chen, Chun, Bin Zhao, and Charles J. Weschler. "Assessing the Influence of Indoor Exposure to “Outdoor Ozone” on the Relationship between Ozone and Short-term Mortality in U.S. Communities." National Center for Biotechnology Information. U.S. National Library of Medicine, 18 Nov. 2011. Web. 12 Dec. 2014. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3279450/>.
Environmental Protection Agency. "Indoor Air Publications: Ozone Generators That Are Sold as Air Cleaners." EPA. Environmental Protection Agency, 2014. Web. 12 Dec. 2014. <http://www.epa.gov/iaq/pubs/ozonegen.html>.
"Ground Level Ozone." EPA. Environmental Protection Agency, 1 Dec. 2014. Web. 09 Dec. 2014. <http://www.epa.gov/groundlevelozone/>.
Norris, Kara. Air Quality Monitoring Lab. N.p.: Web. 2 Dec. 2014. PDF.
"Particulate Matter." EPA. Environmental Protection Agency, 18 Mar. 2013. Web. 10 Dec. 2014. <http://www.epa.gov/pm/>.
"The Regional Impacts of Climate Change." Intergovernmental Panel on Climate Change. IPPC, n.d. Web. 12 Dec. 2014. <http://www.ipcc.ch/ipccreports/sres/regional/index.php?idp=231>.
According to the Environmental Protection Agency, the Clean Air Act has led to economic prosperity in recent years: The United States environmental protection technologies industry, [which includes all environmental media, environmental cleanup, and resource recovery,] by 2007, “was generating approximately $282 billion in revenues, producing $40 billion in exports, and supporting 1.6 million jobs” (Environmental Protection Agency, 2013). In addition, due to the deleterious effects of air pollutants, such as ozone and particulate matter, on public health, a proliferation in air pollution can lead to a significant attrition in the number of workers, which presents negative effects on the economy. In such an unfavorable scenario, air quality monitoring precludes not only harmful effects on the environment, but also the economy. Increased exposure to PM would be harmful to any nation's economy, for there are jobs that need to be carried out. Premature deaths and aggravated respiratory problems disrupt the nation's capitalistic economy by reducing the labor pool in the U.S. and putting people out of work, "as indicated by increased hospital admissions and emergency room visits, school absences, work loss days, and restricted activity days"("Particulate Matter," EPA). Finally, air pollutants, such as sulfur dioxide, can produce acid rain, which kills vegetation, leading to decreased crop yields, and ultimately harming the agricultural industry. From these conclusions, it is important to walk away with one thing in mind: Just as atmospheric conditions and O3 and PM concentrations influence one another, human activities influence the formation of these air pollutants and those pollutants will inevitably affect them.
Citations:
Allen, Jeannie. "National Aeronautics and Space Administration." NASA GISS: Research Features: Ozone and Climate Change. NASA, Feb. 2004. Web. 12 Dec. 2014. <http://www.giss.nasa.gov/research/features/200402_tango/>.
Chen, Chun, Bin Zhao, and Charles J. Weschler. "Assessing the Influence of Indoor Exposure to “Outdoor Ozone” on the Relationship between Ozone and Short-term Mortality in U.S. Communities." National Center for Biotechnology Information. U.S. National Library of Medicine, 18 Nov. 2011. Web. 12 Dec. 2014. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3279450/>.
Environmental Protection Agency. "Indoor Air Publications: Ozone Generators That Are Sold as Air Cleaners." EPA. Environmental Protection Agency, 2014. Web. 12 Dec. 2014. <http://www.epa.gov/iaq/pubs/ozonegen.html>.
"Ground Level Ozone." EPA. Environmental Protection Agency, 1 Dec. 2014. Web. 09 Dec. 2014. <http://www.epa.gov/groundlevelozone/>.
Norris, Kara. Air Quality Monitoring Lab. N.p.: Web. 2 Dec. 2014. PDF.
"Particulate Matter." EPA. Environmental Protection Agency, 18 Mar. 2013. Web. 10 Dec. 2014. <http://www.epa.gov/pm/>.
"The Regional Impacts of Climate Change." Intergovernmental Panel on Climate Change. IPPC, n.d. Web. 12 Dec. 2014. <http://www.ipcc.ch/ipccreports/sres/regional/index.php?idp=231>.